- Home
- en
- datalogger-monitoring-systems
- datatrace-software
DataTrace® - Software
The core of your process monitoring for your MPRF and MPIII data loggers is the intuitive and data-secure DT Pro software, which is compliant with GMP and FDA 21CFR part 11 guidelines and is unsurpassed in terms of user-friendliness and flexibility.
- Create reports in any format
- Save time and money with automatic data capture and charting.
- Utilize password-protected user access, digital signatures and automatically generated audit trail documentation to make your work more efficient.
- Synchronize the programming of multiple loggers in any combination of types
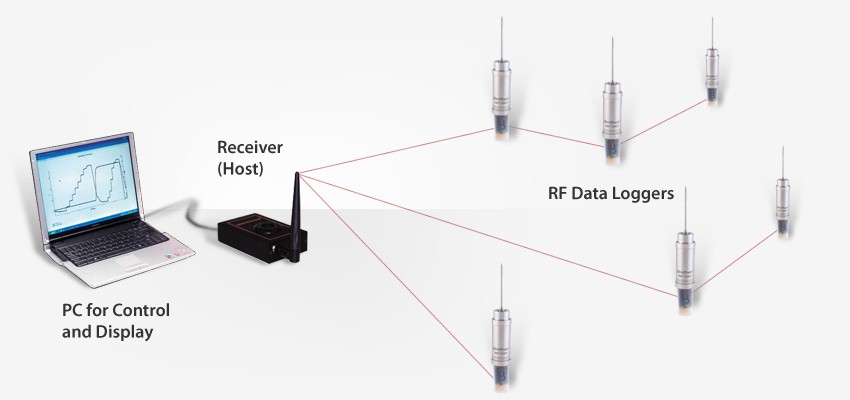
The DT Pro software can be used as stand-alone software or as a network application that leaves nothing to be desired.
License variants
| License variants | 21 CFR Part 11 Compliant | Radio transmission | Validation documentation | License Description |
|---|---|---|---|---|
| DT Pro Basic license |
NO | NO | NO | Includes all tools for programming, reading, testing and calibrating loggers, creating and customizing reports and managing data. |
| DT Pro Plus license |
YES | YES | YES | Standard Edition plus support of FDA 21CFR11, wireless data reception and alarm function. |
| DT Pro LAN Server License |
YES | YES | YES | Plus Edition plus necessary components to use the database with other Data Trace Pro installations. Note: only one LAN Server Edition is required to share the LAN installation; Plus Editions include the components necessary to connect to the LAN Server database. |
3 reasons why you should use the DataTrace software:
- +
Minimal training for maximum productivity
DT Pro software is packed with time-saving features such as keyboard shortcuts (to minimize keystrokes), intuitive dialog boxes and batch functions. Unrivaled reporting capabilities allow you to precisely pre-define which reports are needed, which can then be saved as templates for instant recall when needed. On-site calibration is quick and easy. The battery life of a logger is displayed during programming and can therefore be checked before use.
- +
Unique product features and outstanding data security meet the requirements of the regulatory authorities
DT Pro software combines intelligent data security with regulatory requirements, including an audit trail at all security levels, data encryption, digital signatures, enhanced password protection and administrative security options. Validation of the installation is made even easier with the new system configuration reports. IQ/OQ/PQ documents and a validation manual are also available. All this enables a smooth FDA 21 CFR 11 or GxP compliant implementation
- +
Intuitive, easy-to-use user interface
DT Pro helps you manage your processes, your budget and your time. You will find many features that make your validation and reporting tasks easier in your day-to-day work. Compared to software from other manufacturers, the user interface is more intuitive and has more graphical controls. This leads to better understanding, easier operation and greater effectiveness. Create reports using the simplified report function, analyze data and define your own error conditions. Configure different alarm conditions and email notifications.
When the pressure is on - rely on DataTrace!
These pages may also interest you



