- Home
- en
- datalogger-monitoring-systems
- processvalidation-applications
Our validation loggers in use
Optimization of our loggers with the help of a packaging service provider
Sonoco is a provider of consumer packaging, packaging for industrial products and packaging solutions and services. They use data loggers in various environments and areas of application, including high temperatures and high acid levels. However, the loggers sometimes failed due to water ingress. The following solution was found.
- +
Learn more
Sonoco is a $5 billion global provider of consumer packaging, industrial products, protective solutions and display and packaging services. Headquartered in Hartsville, S.C., with more than 330 operations in 34 countries, they produce packaging for a variety of industries and many of the world's most recognized brands, serving customers in 85 nations.
Sonoco's operations consist of a global consumer packaging businesses -Rigid Paper and Closures, Flexibles and Plastics; their display and packaging division, which provides high-impact retail displays and packaging supply chain management; their industrial businesses, which manufacture tubes and cores, reels and spools, uncoated recycled paperboard and Sonoco Recycling, one of the world's largest recyclers; and their Protective Solutions division, which produces custom-designed protective, temperature-assurance and retail security packaging solutions and highly engineered components.
The Challenge(s)
Sonoco uses Mesa Labs' MPIII data loggers in various environments and applications, including high-temperature and high-acidity. On occasion, it has been noted that data loggers fail for water intrusion. This may sometimes be caused by the flattening of the o-ring.
The Solution(s)
Mesa Labs is committed to continual improvement of our products, as well as increased education to gain the best performance of our products for our customers. We suggest a parameter of 25% battery life be a marker for the battery to be replaced. When customers order new batteries from Mesa for their data loggers, a new o-ring and silicone are automatically included in the order. For each battery replacement, Mesa suggests replacing the o-ring, and applying a thin layer of silicone grease to increase the fluid barrier. We also intend on distributing our simple-to-follow battery replacement instructions with each battery and data logger order. This will provide better instructions for our clients on best practices.
More efficiency and cost savings with validation loggers
A manufacturing company for contract development and production organization supports the medical technology industry with pharmaceutical, biopharmaceutical and medical devices. Our validation loggers ensure flexibility and time savings. They make it possible to restart a validation if the real-time temperatures deviate from the prescribed scale.
- +
Learn more
Case Study: Confidential Client
A cGMP contract development and manufacturing organization, based in Irvine, California, supports the pharmaceutical, biopharmaceutical and medical device industries. The customer's GMP services includes parenteral manufacturing (liquid/lyo), formulation development, analytical chemistry, analytical development, biopharmaceutical development, stability storage, structural chemistry, microbiology, and drug delivery device testing.
The Challenge
The customer has 66 chambers (refrigerators, freezers, incubators and ovens), which require semi-annual validation and qualification of the temperature. Using a competitive logger, they had to wait 24-48 hours to determine if the temperature had been out of specification. The process involved a pre-test of 4 hours, the 24-48 hour IOQ cycle, and then another 4 hour post-test with a competitive data logger. During this cycle, they were unable to tell if the loggers failed or remained within specifications until the full process had been cycled through.
The Solution
The Mesa Labs' MPRF data loggers provided the customer with the flexibility to restart the validation if they see the real-time temperature go out of the specified range. Prior to using the Mesa Labs MPRF data logger, they were using a competitive product without real-time data capability. By using Mesa's MPRF logger, they were able to decrease the cycle time by 12 hours on every chamber! Since 3 chambers are qualified each month, this saves the customer 432 hours of cycle time each year, estimating $8,600 in labor savings alone per year.
Testemonial
"The Mesa Labs MPRF data loggers have allow us to view real-time temperature data and stop and restart the qualification of the cycle if required. This has allowed us to become more efficient and reduce labor cost."
- Installation, Calibration & Validation Services
Validation of freeze-drying processes
Lyophilization is a freeze-drying process in which the product is exposed to a very cold ambient temperature and kept under vacuum. During this process, it is particularly important that the temperature-controlled shelves bring the product to the correct temperature.
Pasteurization of beverage cans
Measurements of temperature and pressure curves inside beverage cans are essential in order to adapt pasteurization on a laboratory scale precisely to the industry. The company Xolutions GmbH, at home in beverage and plastics technology, uses the MPIII data loggers of the DataTrace series for the exact determination of temperature and pressure curves within beverage cans.
Temperature monitoring during retort sterilization
One of the most common methods for sterilizing food is retort sterilization in autoclaves. With our autoclave loggers, you can monitor your temperature data in real time and thus optimize your food production in many areas and provide traceable proof of quality.
Save time with thermal validation
Is relocating thermocouples for your validation tasks becoming too tedious? Do you have difficulties with the correct positioning of thermocouples in the chamber and containers? Have you had enough of the tedious pre- and post-calibration of thermocouples? If you have answered "yes" to even one of these questions, our data logger systems offer you ideal validation solutions!
- +
Learn more
Chamber mapping and heat penetration studies are key to validating your equipment and processes and therefore an important part of your GxP compliance. Our solutions replace thermocouple-based validation solutions and allow you to perform validation in a fraction of the time. With our solutions, you can perform these critical studies in a fraction of the time. In the following example, the thermal mapping of a 16-point autoclave, our DataTrace solutions save 65% time compared to thermocouple systems.
Work step Thermocouples DataTrace Pre-calibration 30 minutes - Placement 30 minutes 5 minutes Measurement 45 minutes 45 minutes Distance 15 minutes 2 minutes Recalibration 30 minutes - Total 150 minutes 52 minutes No need for pre- and post-calibration
Unlike thermocouples, the thermistor and RTD technology used in DataTrace data loggers is stable over a long period of time. This stability allows you to significantly reduce or eliminate the time between calibration checks.
Simplified sensor placement
DataTrace data loggers can be easily and quickly placed in chambers or rooms. Because they are wireless, there's no need to fuss with thermocouples or power cables - just position the loggers in a convenient location and you're ready to collect data.
Improve the accuracy of heat penetration studies
The DataTrace data loggers are extremely small, robust precision instruments that can be easily placed in product packaging. Using appropriate fixtures, the tip of the temperature probe can be positioned precisely at the coldest point within the packaging, providing extremely accurate heat penetration information. The product package can be sealed in its normal process, often without the need to insert "openings" for thermocouple wires into the package.
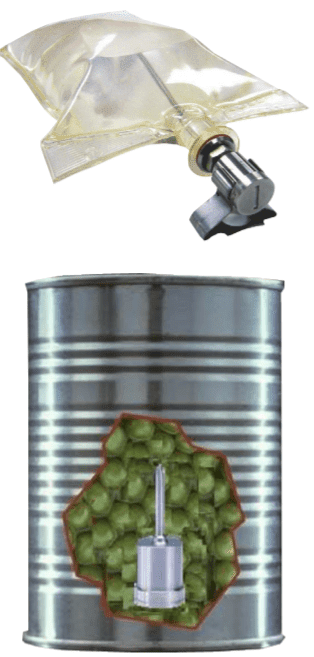
Real-time validation using autoclave loggers
Mesa Laboratories, Inc, has indeed found a completely wireless solution that eliminates the use of thermocouples in autoclave validation.
How can you successfully send an uninterrupted wireless signal from a cascading waterfall autoclave during the sterilization of IV bags (pouch, pouch pouches) or canned food products?
- +
Learn more
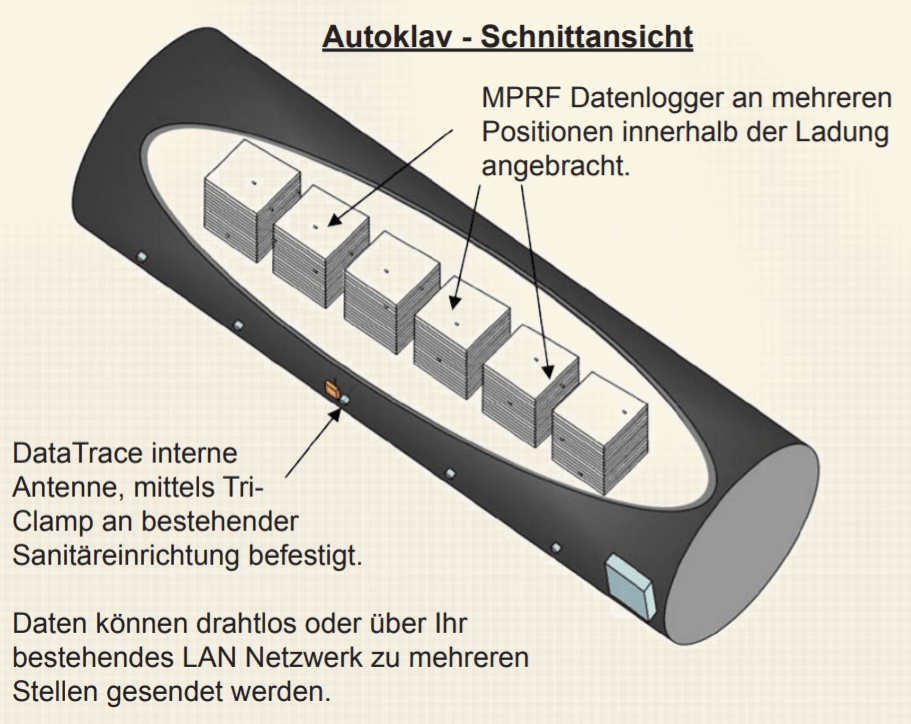
Real-time validation using wireless autoclave loggers eliminates thermocouples in high-volume industrial sterilization of parenterals (infusion bag monitoring, plasma bag monitoring, pouch monitoring)Mesa Laboratories, Inc, has indeed found a completely wireless solution that eliminates the use of thermocouples in autoclave validation. How can you successfully transmit an uninterrupted wireless signal from a cascading waterfall autoclave during the sterilization of IV bags or canned foods? Mesa Labs has developed a prototype for an internal radio antenna* that can be easily attached to existing sanitation equipment using a Tri-Clamp connector. This allows operators to quickly and easily convert the antenna from one autoclave to another without the use of special tools. Once the antenna is fitted, data can be analyzed via the DataTrace Pro software on a laptop near the autoclave, in the control room using an RF repeater or, when using DataTrace Pro LAN, even anywhere where a PC with a network connection is available.

Improved report options

Increased graphical control
DataTrace Pro uses an SQL database that is accessible from other programs and will soon replace thermal recorders and other thermocouple-based systems. Mesa Labs introduces a completely new concept in data loggers with the DataTrace MPRF. Even if radio contact is lost, your data will not, and DataTrace Pro will automatically recover it.DataTrace Pro software is specifically designed for engineers and technicians in the validation industry working in the food and pharmaceutical industries. Some outstanding features include:
- Wireless, fully automated verification through pre- and post-calibration with unlimited number of calibration points, using your in-house calibration facilities
- Fully configurable and customized reports
- User-configurable statistical evaluation; users can assign their own acceptance criteria
- Fully CFR Part 11 compliant - developed in a GmP environment for GxP applications
- Real-time alarming with user-definable options and e-mailing of alarm conditions
- Easy LAN configuration
- +
Learn more
DaTrace data logger in smokehouses
Demonstration for management and plant quality control teams of a meat product manufacturer to specifically demonstrate MPRF and DTRF models in use in 15 smoking chambers. The smokehouses where the MPRF loggers were deployed were at least 45 meters away from an enclosed control room where DTRF was running.
Challenge & Solution
Currently, Quality Control sends a technician into the smokehouse once the cooking time is reached to take a direct temperature measurement with a NIST traceable thermometer. The technician must take at least six different measurements inside each smokehouse. The smokehouses are HOT, SMOKING, FULLY PACKED with product and DANGEROUS.
The transmission of live data from the smoking chambers greatly reduces the workload for the technician responsible for recording the temperature. It also enables the system to better meet certain requirements, such as those imposed by HACCP and other safety and quality initiatives.
The MPRF loggers were tested in the smoking chambers furthest away (approx. 45 meters) from the control room. The QC manager then placed the MPRF units at the rear of the chambers (a further 6 meters away). Two repeaters were positioned in a direct line between the control room and the smoking chambers. Their placement was critical to relay the radio signal to the control room where DTRF was running on a PC.
A laptop and PC interface were deployed with normal antenna configuration outside the control room and achieved 100% recovery of live data.
- +
Learn more
Study on heat penetration in medical hazardous goods
Equipment: 5 data loggers per autoclave - 4 penetration, 1 outlet Exposure time: 30 minutes at 121˚C Cycle: 2 hours The customer chose DataTrace RF for this application for the following reasons:
- The ability to monitor the autoclaves from a laptop in the disposal area or a desktop PC connected to the network.
- N.I.S.T traceable temperature readings in real time from the coldest point in the hazardous material containers.
- The ability to release the batch immediately once all samples have reached the required temperature and exposure time.
- To be able to verify that sterilization has been carried out successfully.
- To reduce the current cycle from 120 minutes to 90 minutes, increasing the efficiency of the autoclaves by 33%.
- To completely eliminate the need for wired sensors, as thermocouples or other systems that rely wholly or partially on wired sensors would dramatically increase load and unload times, negating the time gained by reducing the cycle.
- To avoid validation by expensive third-party vendors if the specified sterilization criteria were not met during the 14-day biological monitoring tests.
- To eliminate the use of chart recorders.
Further applications
Navigation
Produkte
Branchen & Anwendungen

